More Breaking News: U.S. FDA Approves Nemolizumab (Nemluvio, Galderma) for Patients with Moderate to Severe AD
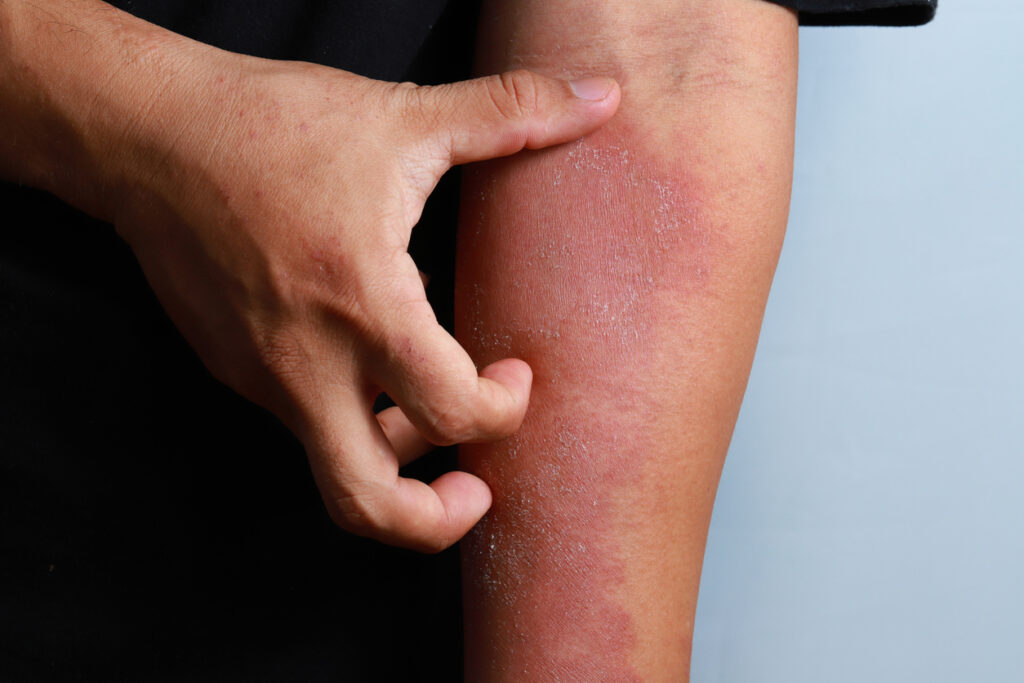
The U.S. Food and Drug Administration (FDA) has approved nemolizumab (Nemluvio, Galderma) for the treatment of patients aged 12 and older with moderate to severe atopic dermatitis, in combination with topical corticosteroids (TCS) and/or calcineurin inhibitors (TCI) when the disease is not adequately controlled with topical prescription therapies. This action follows the recent FDA approval of […]
